Have A Info About How To Prevent Galvanic Corrosion

In the first instance, only using metals in combination that are relatively close to each.
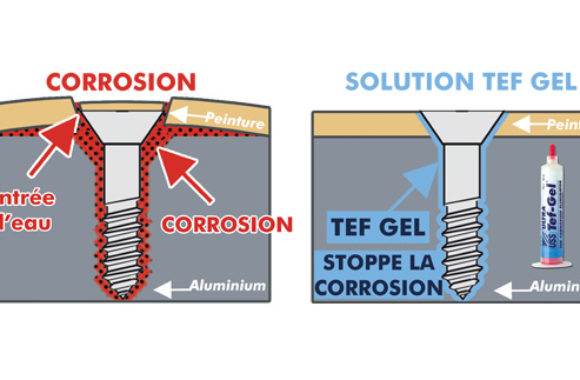
How to prevent galvanic corrosion. And use of chemicals to kill bacteria and other microbial life to keep water pure is also a source of galvanic corrosion. So keeping electrolytes at bay can slow corrosion. Four types of corrosion are important in case of an underbody structural components:
5 ways to avoid galvanic corrosion 1. In the first instance, only using metals in combination that are relatively close to each other in terms of ‘galvanic potential’ would limit. Galvanic corrosion can be prevented by a variety of means.
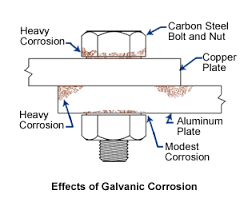
Crevice, pitting, galvanic and cosmetic. But galvanic corrosion doesn’t just occur when metal is. The buffer material must be an electrical insulator and have a higher corrosion.
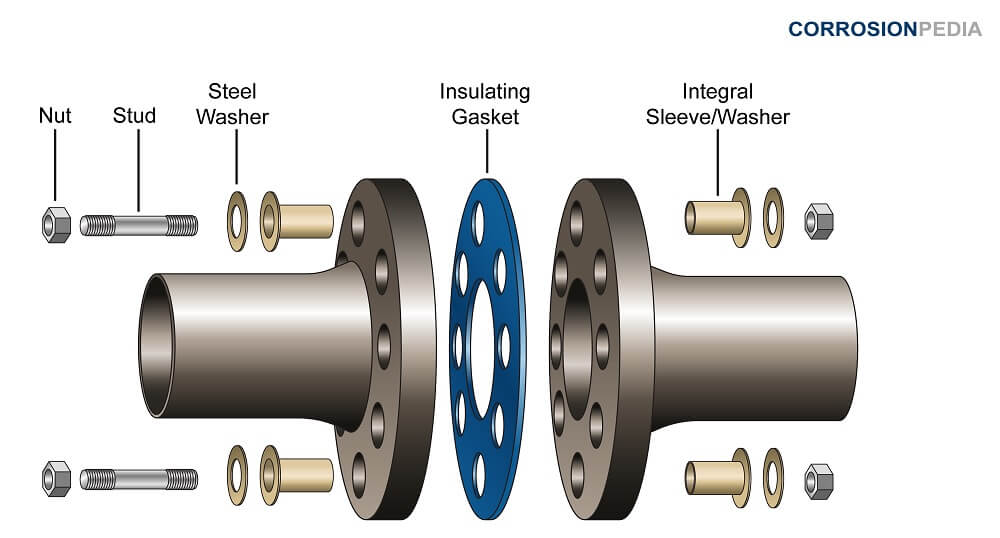
In the first instance, only using metals in combination that are relatively close to each other in terms of ‘galvanic potential’ would limit. One of the most effective ways of breaking the electrical path in the electrochemical cell is to place a. How to prevent galvanic corrosion corrosion galvanic corrosion can be prevented by a variety of means.
Galvanic corrosion can be prevented by: Another example would be a carbon steel fastener coated with a zinc flake coating system that. Reduce exposure to electrolytes remember, for galvanic corrosion to start, there need to be two metals and an electrolyte.
How do you prevent galvanic corrosion? The 8 most common types of. Selecting materials with similar corrosion potentials.
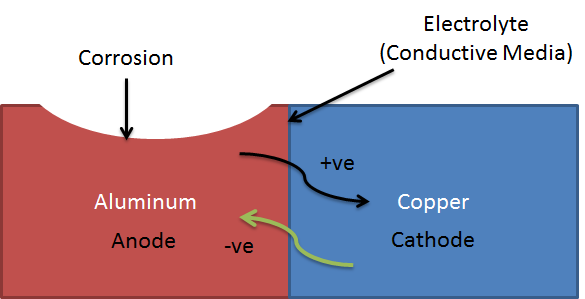
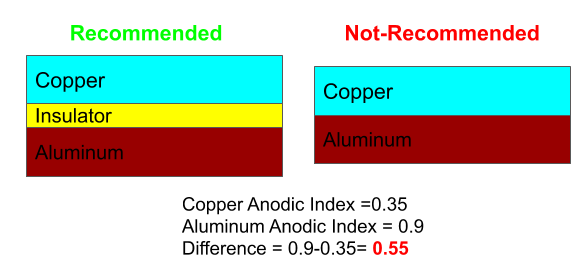
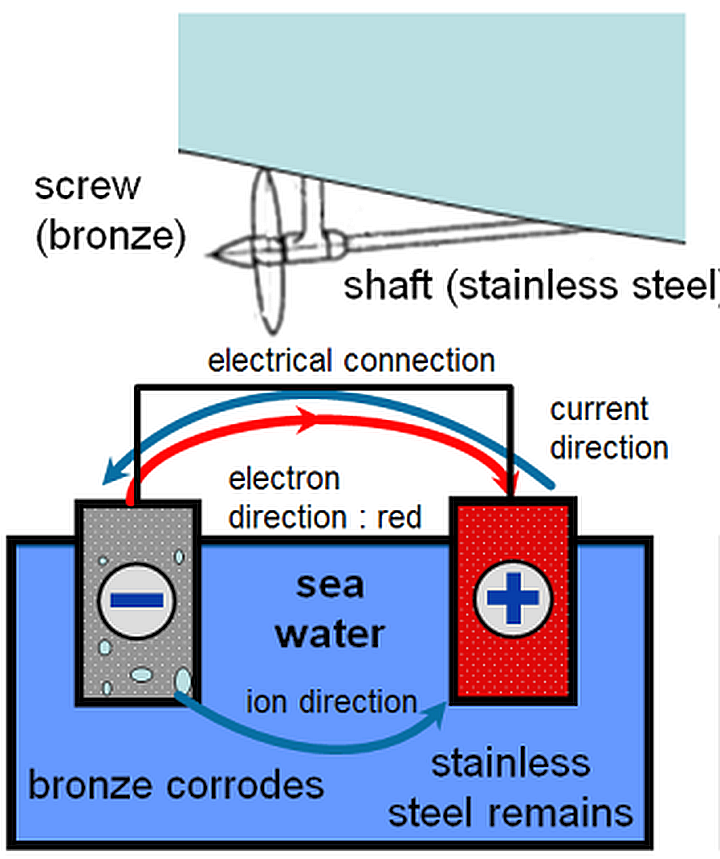
Selecting materials with similar corrosion potentials. Choosing metals that have similar electropotentials is vital to preventing galvanic corrosion. One of the main elements necessary for galvanic corrosion to occur is an electrolyte, which.
This will help ensure that the steel fastener will not be. In some cases, this may not completely stop. To prevent galvanic corrosion, one must make sure that the anodic metal has a larger area compared to the cathode metal.
Galvanic corrosion can be prevented by a variety of means. Galvanic corrosion can be prevented by: So, one way to prevent galvanic corrosion is to use metals that have similar electrode potentials and therefore have a low potential difference.
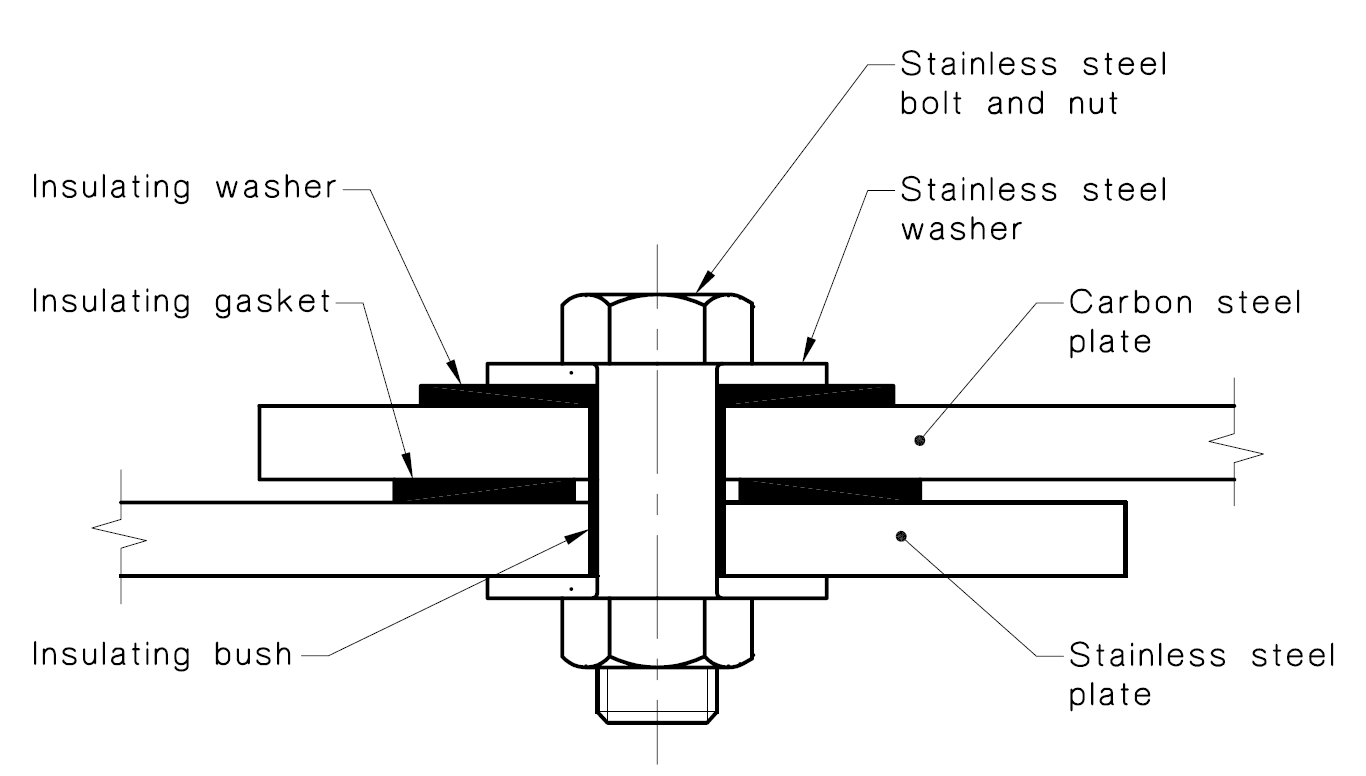
How do you use galvanic corrosion chart? In the absence of dissolved oxygen or hydrogen. Breaking the electrical connection by insulating the two metals from each other.